|
A new measure of ligand field strength has been developed using the variable spin states of iron(III) porphyrins as a function of axial ligand X in FeX(TPP). Contrary to expectations based on the Spectrochemical Series, H 2 O is found in the Magnetochemical Series to be a significantly weaker field ligand than OH - . The full ranking is Ag(CB 11 H 12 ) 2 - < CB 11 H 12 - < SbF 6 - < Co(C 2 B 9 H 11 ) 2 - < AsF 6 - < H 2 O < ClO 4 - < C(CN) 3 - < CF 3 SO 3 - < BF 4 - < ONC(CN) 2 - < ReO 4 - < OTeF 5 - < I - < Br - < OH - ~ Cl - < NCS - < OAc - ~ N 3 - < OPh( p -NO 2 ) - < OPh - < F - < RS - .
This order reflects metal-ligand  bonding whereas the Spectrochemical Series is dominated by bonding whereas the Spectrochemical Series is dominated by  bonding effects. The magnetochemical ranking is suggested to be more useful for correlating ligand field strengths in bioinorganic chemistry. See Evans et al. J. Am. Chem. Soc . , 2000 , 122 , 4660-4667 . bonding effects. The magnetochemical ranking is suggested to be more useful for correlating ligand field strengths in bioinorganic chemistry. See Evans et al. J. Am. Chem. Soc . , 2000 , 122 , 4660-4667 .
Currently, we are exploring the use of 19 F and 13 C NMR spectroscopy to extent and consolidate the magnetochemical Series. 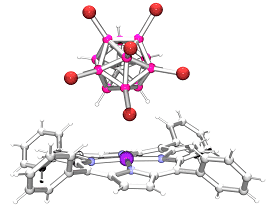
The structure of the nearly "bare" Fe(TPP) + cation having the weakest
axial ligand field in the pure S = 3/2 intermediate spin complex Fe(TPP)(CHB 11 H 5 Br 6 ). [back to projects]
|